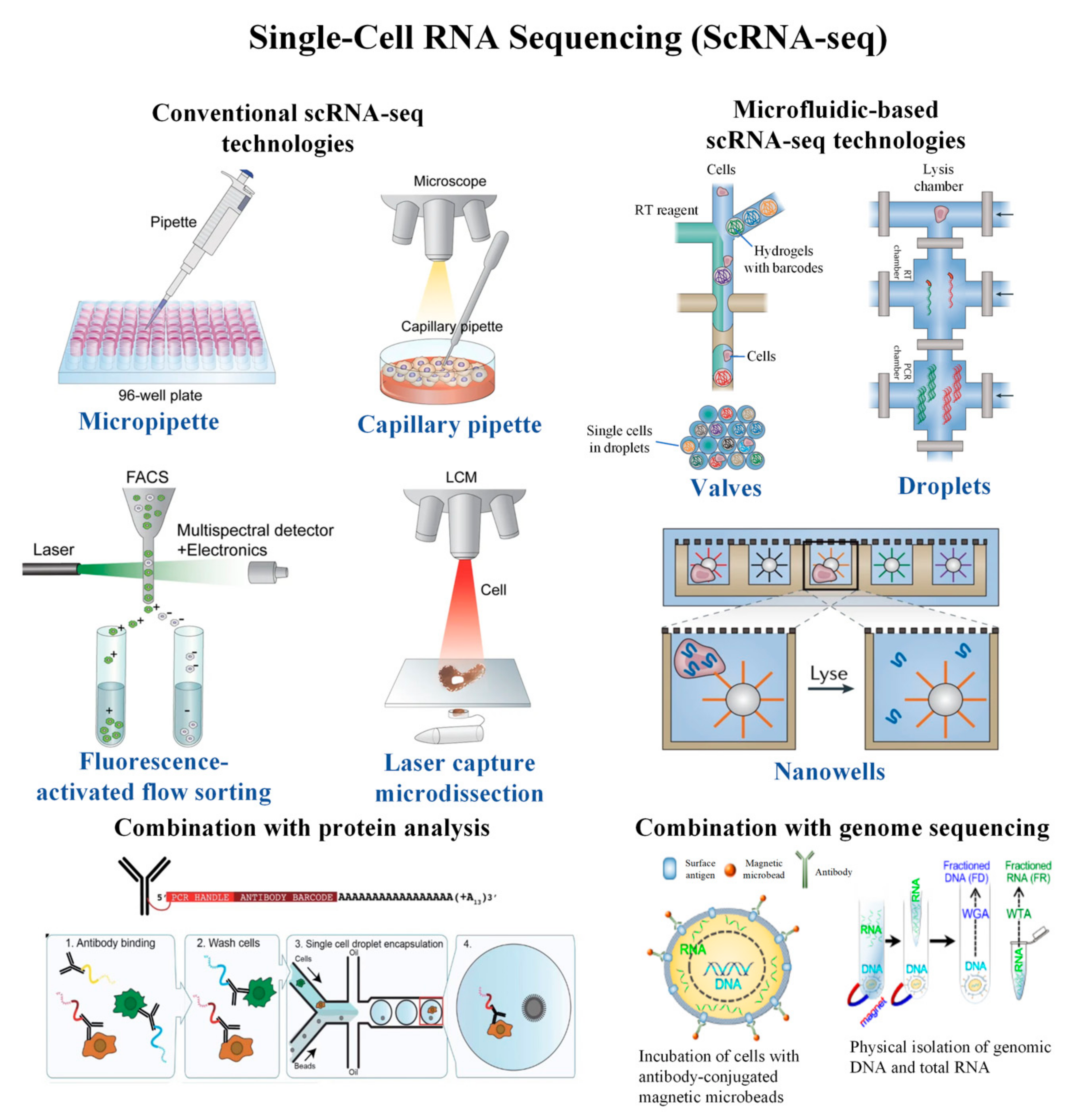
This custom method allows to sequence a large number of cells at low costs and can be adapted to specific questions. Subsequently, droplets are pooled and cDNA is further processed for sequencing. Cells are lysed and poly(A)+ mRNAs are barcoded in the droplets. In this approach, single cells from a cell suspension are isolated into droplets. The Custom Single Cell Omics (CSCO) platform currently develops an inDrop 3’ scRNA-seq method. Effects of CRISPR perturbations can also be assessed at single cell resolution, with direct capture of cellular sgRNAs and changes in gene expression.ģ’ scRNA-seq at the Custom Single Cell Omics platform Using feature barcodes, 3’ scRNA-seq can be combined with cell surface proteins expression using specific antibodies at the single cell level (CITEseq) or to increase the multiplexing capacity (Cell Hashing). The sequencing recommendations are around 50-100k reads per cell or nucleus. The required input material is 1,000 to 20,000 cells or nuclei and the recovery rate (yield) is around 50% of loaded cells/nuclei. 3’ scRNA-seq represents a relatively easy way to quantify the poly(A)+ mRNAs of single cells.ģ’ scRNA-seq is available in routine at the NGS platform using the 10X Genomics pipeline. Only the 3’ ends of the transcripts bear the barcodes, are represented in the final libraries and will be sequenced. The generated cDNA molecules are amplified and then fractionated. This polydT sequence is located directly adjacent to the barcodes required for the identification of unique cells and for sequencing. In 3’ scRNA-Sequencing, poly(A)+ mRNAs are reverse transcribed using a polydT primer. 3’ RNA sequencing allows to quantify gene expression and to identify cell populations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
